Psychedelic Therapies Evolving with Patented Improvements
Robin Lefferts
January 6th, 2022
News, Psychedelics, Top News
Psychedelic substances, such as psilocybin and DMT, have shown great promise in the treatment of a variety of psychiatric disorders, including depression, PTSD, and anxiety. The findings are driven by a wide range of studies, including from esteemed institutions such as Johns Hopkins and the Imperial College of London. In fact, the US National Institutes of Health recently agreed to fund studies conducted by Johns Hopkins to investigate whether psilocybin can help people quit smoking. The news is very promising.
But, of course, there is a caveat. Psychedelic therapies typically have extremely long durations and can have undesirable side-effects. They usually have to be guided by medical professionals in a clinical environment, which means that a long duration creates capacity constraints that could prevent these therapies from being scalable.
Mindset Pharma Inc. (CSE: MSET) (OTCQB: MSSTF) is addressing these safety and duration concerns by developing a broad array of next generation psychedelics. These molecules are inspired by the natural compounds but are designed to improve their performance in a variety of ways. The company’s approach has an added benefit, in that its drugs can be patented, unlike the historical psychedelics that reside in the public domain. It takes a lot of research and development (read time and money) to bring a drug through the clinical trial process, and Mindset is betting the appetite for IP-protected treatments will be robust compared to those that can’t be protected.
How and why was Mindset Pharma started?
Next Generation Psychedelics Are Here
Mindset Pharma has, so far, synthesized over 100 novel compounds, inspired by and modeled after existing psychedelics. The company has also filed eight patent applications, covering a broad range of next generation drugs as well as a novel method of synthesizing psilocybin. Mindset’s drugs fall broadly into four ‘families’ of molecules, according to their specific actions.
Family 1 is psilocybin-inspired, and the lead candidate has the potential to be stronger, safer, and more cost-effective than the original model. Preclinically, MSP-1014 has demonstrated a significantly improved safety profile in preclinical studies, including completely attenuating the reduction in core body temperature that was observed with psilocybin. It also shows a stronger effect on the 5-HT2A receptors that psilocybin targets. It’s designed to potentially replace psilocybin and could require lower dosage levels to achieve the same effects.
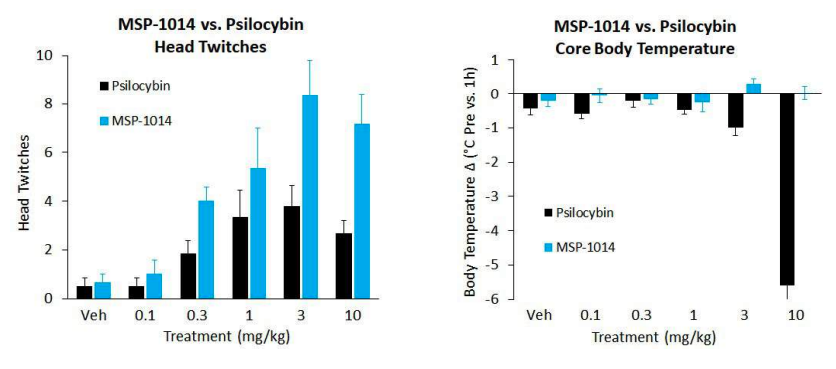
Source: Mindset Pharma Corporate Presentation
Family 2 is similar to Family 1, except the molecules in this family have a shorter duration of effect than psilocybin does. They could be suitable in cases where macrodosing in a clinic is called for but would offer the advantage, to both the clinic and the patient, of a much shorter visit.
Family 3 consists of potential microdosing solutions suitable for daily, at-home treatments. These molecules offer a lower but more sustained potency than their psilocybin ‘parents.’ They may not require a long psychedelic trip to provide benefit, instead providing a more constant drip of the desired receptor activity.
Family 4 leaves the world of psilocybin and is instead inspired by 5-MeO-DMT. DMT itself is perhaps more widely known as an element of shamanic ayahuasca rituals and is naturally occurring. It has a long duration and delivers intense, visual, out-of-body types of experiences. 5-MeO-DMT is also naturally occurring and can be synthesized as well. It delivers a much shorter but perhaps more intense psychedelic experience. This intensity can lead to several undesirable side effects. Preclinically, Mindset’s versions of 5-MeO-DMT appear to provide similar benefit with greatly reduced side effects.
Mindset’s Business Model
How does Mindset Pharma plan to monetize its proprietary molecules?
Though there is a significant amount of scientific research that goes into Mindset Pharma’s business, the model for making money is fairly straightforward and on the verge of being implemented. The first step is of course the discovery, design, and testing of potential next generation psychedelic molecules. For some of the company’s current candidates that process is nearly complete.
Armed with promising preclinical data, the company can enter the next phase which is commercialization. Mindset Pharma has no illusions that it can bring a drug candidate through the clinical trial process all the way to the market. The company’s expertise lies in drug discovery. So the plan is to partner with more established pharmaceutical companies and research organizations with those capabilities. Those partnerships could come in a variety of forms, from potentially selling the molecule outright, to licensing its use for a particular application or co-development and collaboration agreements. There are any number of ways to structure such deals, from upfront payments to funding agreements to ongoing royalties and more.
What kind of developments could Mindset Pharma see in the next year or so?
Having built the foundation, with over 100 molecules and four families of drugs currently under its belt, Mindset Pharma anticipates a robust commercialization phase beginning in 2022. The company is also constantly building its patent-pending portfolio, branching out into new areas of research outside of its current psilocybin- and 5-MeO-DMT-inspired creations.
Mindset recently started the commercialization ball rolling, announcing a collaboration with The McQuade Center for Strategic Research and Development. The McQuade Center is part of the Tokyo-based multinational company Otsuka Pharmaceutical, and is dedicated to advancing and nurturing early-stage opportunities that can change the landscape of treatments for mental illness and renal disorders. The McQuade Center has made a strategic investment in support of Mindset’s research, with an upfront payment of $5 million, and is committed to financing two families of Mindset compounds through the completion of Phase 1 clinical trials. In return, The McQuade Center gets a right of first refusal on drugs developed under the collaboration.
The Upshot
There is much more to discuss with Mindset Pharma and we will be doing just that. For now, keep an eye on the company as it moves into commercialization and continues innovating in what amounts to a brand-new industry. With an estimated 280 million people around the world suffering from depression, and effective treatments for the disease largely lacking, Mindset Pharma has the potential to be in just the right place, at just the right time.
This article was published by CFN Enterprises Inc. (OTCQB: CNFN), owner and operator of CFN Media, the industry’s leading agency and digital financial media network dedicated to the burgeoning CBD and legal cannabis industries. Call +1 (833) 420-CNFN for more information.
Network Partners
About CFN Media Group
CFN Enterprises Inc. (OTCQB: CNFN) owns and operates CFN Media Group, the premier agency and financial media network reaching executives, entrepreneurs and consumers worldwide. Through its proprietary content creation, video library, and distribution via www.CannabisFN.com, CFN has built an extensive database of cannabis interest, assisting many of the world’s largest cannabis firms and CBD brands to build awareness and thrive. For more information, please visit www.cfnenterprisesinc.com.
Disclaimer: Matters discussed on this website contain forward-looking statements that are subject to certain risks and uncertainties that could cause actual results to differ materially from any future results, performance or achievements expressed or implied by such statements. CFN Media Group, which owns CannabisFN, is not registered with any financial or securities regulatory authority and does not provide nor claims to provide investment advice or recommendations to readers of this release. CFN Media Group, which owns CannabisFN, may from time-to-time have a position in the securities mentioned herein and will increase or decrease such positions without notice. The Information contains forward-looking statements, i.e. statements or discussions that constitute predictions, expectations, beliefs, plans, estimates, or projections as indicated by such words as "expects", "will", "anticipates", and "estimates"; therefore, you should proceed with extreme caution in relying upon such statements and conduct a full investigation of the Information and the Profiled Issuer as well as any such forward-looking statements. Any forward looking statements we make in the Information are limited to the time period in which they are made, and we do not undertake to update forward looking statements that may change at any time; The Information is presented only as a brief "snapshot" of the Profiled Issuer and should only be used, at most, and if at all, as a starting point for you to conduct a thorough investigation of the Profiled Issuer and its securities and to consult your financial, legal or other adviser(s) and avail yourself of the filings and information that may be accessed at www.sec.gov, www.pinksheets.com, www.otcmarkets.com or other electronic sources, including: (a) reviewing SEC periodic reports (Forms 10-Q and 10-K), reports of material events (Form 8-K), insider reports (Forms 3, 4, 5 and Schedule 13D); (b) reviewing Information and Disclosure Statements and unaudited financial reports filed with the Pink Sheets or www.otcmarkets.com; (c) obtaining and reviewing publicly available information contained in commonlyknown search engines such as Google; and (d) consulting investment guides at www.sec.gov and www.finra.com. You should always be cognizant that the Profiled Issuers may not be current in their reporting obligations with the SEC and OTCMarkets and/or have negative signs at www.otcmarkets.com (See section below titled "Risks Related to the Profiled Issuers, which provides additional information pertaining thereto). For making specific investment decisions, readers should seek their own advice and that of their own professional advisers. CFN Media Group, which owns CannabisFN, may be compensated for its Services in the form of cash-based and/or equity-based compensation in the companies it writes about, or a combination of the two. For full disclosure, please visit: https://www.cannabisfn.com/legal-disclaimer/. A short time after we acquire the securities of the foregoing company, we may publish the (favorable) information about the issuer referenced above advising others, including you, to purchase; and while doing so, we may sell the securities we acquired. In addition, a third-party shareholder compensating us may sell his or her shares of the issuer while we are publishing favorable information about the issuer. Except for the historical information presented herein, matters discussed in this article contain forward-looking statements that are subject to certain risks and uncertainties that could cause actual results to differ materially from any future results, performance or achievements expressed or implied by such statements. CFN Media Group, which owns CannabisFN, is not registered with any financial or securities regulatory authority, and does not provide nor claims to provide investment advice or recommendations to readers of this release. CFN Media Group, which owns CannabisFN, may from time to time have a position in the securities mentioned herein and will increase or decrease such positions without notice. For making specific investment decisions, readers should seek their own advice and that of their own professional advisers. CFN Media Group, which owns CannabisFN, may be compensated for its Services in the form of cash-based and/or equity- based compensation in the companies it writes about, or a combination of the two. For full disclosure please visit: https://www.cannabisfn.com/legal-disclaimer/.
Copyright © Accelerize Inc. · All Rights Reserved · Privacy Policy · Legal Disclaimer